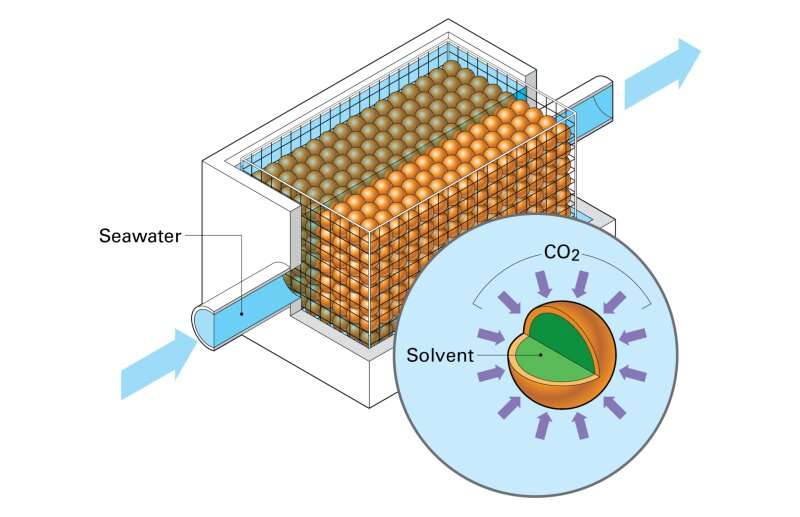
Encapsulated solvent. Source: Rick Henkel
The ocean is not only affected by climate change – it can also be part of the solution to its change.
Direct Ocean Carbon Capture (DOC) is a new form of negative emissions technology that has advantages over its on-land counterpart, direct air capture, due to its ability to avoid land use. DOC can also be easily assimilated by the coastal wind and store carbon dioxide on the coast.
Katherine Hornbostel, an assistant professor of mechanical engineering and materials science at the University of Pittsburgh Swanson School of Engineering, is well versed in the field of carbon capture technologies. He is actively collaborating with Assistant Professor Tagbo Niepa from Pitt’s Department of Chemical and Petroleum Engineering to develop new solutions to capture carbon in the oceans.
The team published two sister papers, “Demonstration of direct ocean carbon capture using encapsulated solvents” and “Demonstration of direct ocean carbon capture using hollow fiber membrane contactors,” in Journal of Chemical Engineering. These two papers show experimentally and computationally how two types of membrane contactors-encapsulated solvents and hollow fiber membrane contactors-can capture carbon dioxide from the ocean.
“Membrane contacts are just what they sound like,” Hornbostel said. “They are membranes that bring two fluids in contact with each other. In this case, we bring together sea water on one side and a solvent on the other.”
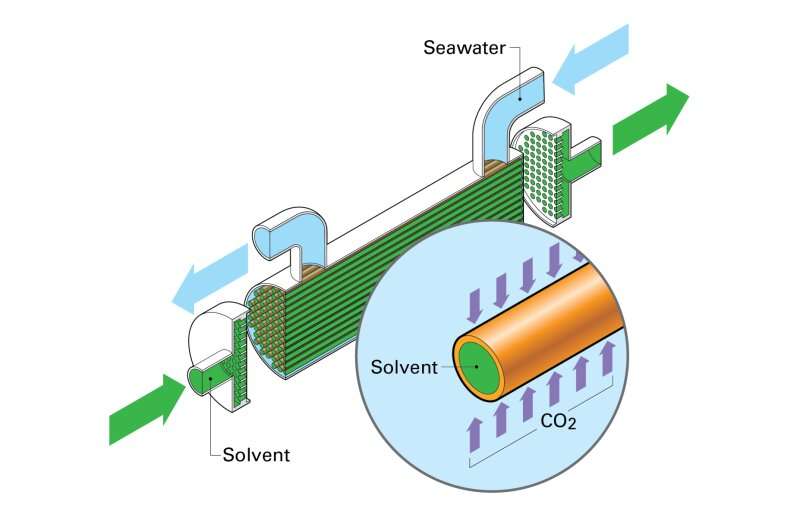
Hollow Fiber. Source: Rick Henkel
The team tested two types of membrane contactors: hollow fiber and encapsulated solvents. The biggest difference between the two technologies is their form. While hollow fiber membrane contactors look like straws, encapsulated solvents look like caviar. Otherwise, they work the same.
“The idea of both is to get a high contact area between seawater and solvent,” explained Hornbostel. “The more surface area you have, the better the rate of carbon dioxide removal.”
Shake the sea water
The carbon dioxide wants to travel across the membrane into the solvent, which is made from the sodium solution that reacts with the carbon dioxide. When seawater comes into contact with the solvent, the carbon dioxide reacts and separates from the seawater. The solution needs to be recirculated to make the process more cost-effective—something the team is still working to improve.
“Theoretically, we could change the price if we could change the pH of the seawater side,” Hornbostel said. “Carbon dioxide is not normally available in seawater at the baseline pH level, so you have to move the pH lower in the seawater to make it more acidic and then more carbon dioxide bubbles.”
Hornbostel’s team is currently looking for ways to swing the pH of seawater using membrane surface treatments and investigating coupling direct seawater capture with desalination to lower system costs.
More information:
Austin Lieber et al, Demonstration of direct ocean carbon capture using encapsulated solvents, Journal of Chemical Engineering (2023). DOI: 10.1016/j.cej.2023.144140
Joanna Rivero et al, Demonstration of direct ocean carbon capture using hollow fiber membrane contactors, Journal of Chemical Engineering (2023). DOI: 10.1016/j.cej.2023.143868
Provided by the University of Pittsburgh
Citation: Using our oceans to fight climate change (2023, July 24) retrieved on July 24, 2023 from https://techxplore.com/news/2023-07-oceans-climate.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
