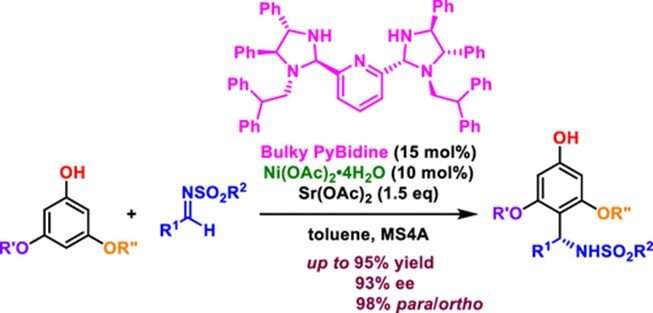
A new bulky PyBidine-Ni(OAc)2 The catalyst developed by Chiba University researchers expands the range of aza-Friedel−Crafts reactions by favoring the production of para-substituted products over conventional ortho-substituted products, opening up new possibilities for the creation of a variety of novel compounds. Credit: ACS Catalysis
From life-saving drugs and synthetic polymers to various advanced materials, the products containing organic compounds seem endless, thanks in part to regioselectivity, a part of chemical reactions in which a substituent is chosen to be added to a specific position in an organic compound. It favors the formation of desired products with specific uses.
A unique regioselective reaction used for the precise design of organic compounds is the Friedel−Crafts reaction, which enables the addition of substituents to specific positions of aromatic compounds such as benzene and phenol rings.
In the traditional acid-catalyzed aza-Friedel-Crafts reaction of phenols with imines, the substitution reaction usually occurs next to the hydroxy (-OH) group of the phenol ring. This leads to the formation of ortho-products such as α-aminobenzylphenols, which are of great importance in the production of agrochemicals and medicines. However, achieving selective substitution at the para-position further away from the -OH is challenging and has only been observed on a few substrates.
In a new study published in the journal ACS Catalysis, Professor Takayoshi Arai from the Graduate School of Science at Chiba University in Japan, together with Masters students, Mr. Ryoya Tajima and Mr. Takaaki Saito, made a significant breakthrough by developing a general para-selective aza-Friedel−Crafts type reaction of phenols with imines. Their work opens up new possibilities for the rapid and safe synthesis of drugs and advanced materials.
“There is no general way to achieve a para-selective aza-Friedel−Crafts reaction with phenols until now,” says Prof. Arai, highlighting recent work today.
In the traditional aza-Friedel−Crafts reaction, the ortho-products are formed in the presence of a Lewis acid catalyst. The challenge facing the researchers is to guide the reaction in favor of products that are substituted for para. To solve this issue, they took inspiration from their previous experiments, where they observed that the bis(imidazolidine)pyridine (PyBidine)-metal complex platform has the potential to perform the aza-Friedel−Crafts reaction of phenols with imines due to its ability to act as an acid and a basic catalyst.
In a series of tests, the researchers found in their current work that PyBidine-Ni(OAc)2 complex shows a preference for para-substituted products. They then replaced the benzyl substituent of PyBidine with a bulky diphenylethyl group to obtain a “bulky PyBidine”-Ni(OAc)2 catalyst, which facilitates para-selective aza-Friedel−Crafts reactions with up to 99:1 para/ortho selectivity.
In addition, 3,5-dialkoxyphenols with various sulfonylaldimines give para-substituted products-with up to 93% enantiomeric excess (chiral purity)-in the presence of a Sr(OAc)2 add up.
Using DFT calculations, the researchers found that this regioselectivity is due to a cooperative activation of the highest occupied molecular orbital (HOMO) of 3,5-dialkoxyphenols and the lowest unoccupied molecular orbital (LUMO) of sulfonylaldimines.
The reaction begins with the formation of a nickel phenoxide, which enhances reactivity in the para-position of the phenol group in a process called HOMO activation. The sulfonylaldimine is then attached to this position via hydrogen bonds to the bulky PyBidine-Ni(OAc)2 catalyst by activating the LUMO, resulting in the formation of the final product.
This HOMO-LUMO activation is the key to switching regioselectivity for designing and developing molecules that can be used for drugs, agrochemicals, and other materials.
Indeed, this study is an important step in the field of regioselective reactions for future applications in various industries. “The fast and safe synthesis method has improved the synthetic efficiency of an orexin antagonist and is expected to contribute to an environmentally friendly and sustainable chemistry,” concluded Prof. Arai.
More information:
Ryoya Tajima et al, Asymmetric Para-Selective Aza-Friedel–Crafts Reaction of Phenols Catalyzed by Bulky PyBidine-Ni(OAc)2, 2013-2014. ACS Catalysis (2023). DOI: 10.1021/acscatal.3c01961
Provided by Chiba University
Citation: New study expands scope of aza-Friedel–Crafts reactions (2023, July 20) retrieved 20 July 2023 from https://phys.org/news/2023-07-scope-aza-friedelcrafts-reactions.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
