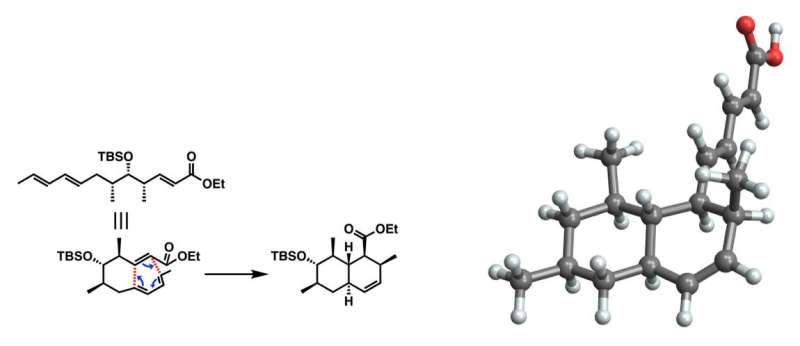
Researchers from TUS, Japan synthesized tanzawaic acid B, a promising antibiotic drug candidate, on the gram scale, artificially for the first time by using intramolecular Diels-Alder reaction, asymmetric alkylation, and asymmetric Mukaiyama aldol reaction. Credit: Isamu Shiina from TUS, Japan
The discovery of antibiotics in 1928 was a major turning point in the history of medicine. For the first time since the dawn of human civilization, doctors have gained access to an extremely powerful and effective tool to fight against a wide variety of bacterial infections. Today, bacterial diseases that used to be a death sentence can be cured, and infections after surgery or chemotherapy can be prevented or treated more effectively.
Unfortunately, the worldwide use (and abuse) of antibiotics has led to the emergence of drug-resistant bacterial strains. Over time, bacteria that would normally be killed by a given antibiotic produce mutant offspring that are immune to it. These mutant strains are a major threat to public health, and the only safe course of action is to develop new antibiotic compounds.
Against this backdrop, a research team including Professor Isamu Shiina, Assistant Professor Takatsugu Murata and Mr. Hisazumi Tsutsui from Tokyo University of Science (TUS) in Japan has now achieved great success in the synthesis of new antibiotics. As reported in their paper published in ACS Omega in July 2023, the team achieved the first ever gram-scale synthesis of tanzawaic acid B, which may serve as a candidate for the discovery of new drugs.
But what is tanzawaic acid B? In 1997, Professor Daisuke Uemura and colleagues working in the Tanzawa area of Japan isolated a series of organic polyketide compounds from the fungus Penicillium citrinum. These compounds are grouped into what we now call the “tanzawaic” acid family, which has several members from A to Z.1.
Tanzawaic acid B has attracted the most attention because it has a common core structure with many tanzawaic acids, which means that an artificial synthesis method for tanzawaic acid B can easily lead to synthesis methods. for others.
However, synthesizing tanzawaic acid B from scratch is a challenging endeavor. Tanzawaic acids feature a polysubstituted octalin skeleton—a structure composed of 10 carbon atoms in a tight pattern with multiple chemical groups at specific locations. The researchers synthesized this skeleton by using a chain-like molecule that they synthesized in a previous study. Then, by using a carefully controlled intramolecular Diels-Alder reaction, they made these chains preferentially “fold” into the desired octalin skeleton.
The next challenge is to precisely rearrange the octalin skeleton in several steps to produce tanzawaic acid B. As octalin has eight carbon atoms that can participate in stereochemical reactions, any desired substitution effectively competes with 255 other possible arrangements.
To solve this issue, the researchers used asymmetric alkylation and asymmetric Mukaiyama aldol reaction, which enabled them to produce the desired polysubstituted octalin compound tanzawaic acid B on a gram scale.
Overall, this new synthesis technique may play a key role in the development of antibiotic drugs based on tanzawaic acids. Prof. Shiina said, “For more than 25 years since its discovery, the total synthesis of tanzawaic acid B has not been realized, until now. new antibiotic candidates for multidrug-resistant bacteria.”
With a continuous, large supply of tanzawaic acids, researchers will soon be able to test their interesting biological activities, including antibacterial, antimalarial, and antifungal properties. “Further developments in the synthesis of tanzawaic acid B are currently underway, along with investigations into its biological activity and synthetic analogues,” concluded Prof. Shiina.
More information:
Takatsugu Murata et al, First Total Synthesis of Tanzawaic Acid B, ACS Omega (2023). DOI: 10.1021/acsomega.3c03634. pubs.acs.org/doi/10.1021/acsomega.3c03634
Provided by Tokyo University of Science
Citation: First artificial synthesis of tanzawaic acid B may lead to new antibiotics (2023, July 19) retrieved 19 July 2023 from https://phys.org/news/2023-07-artificial-synthesis-tanzawaic- acid-antibiotics. html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
