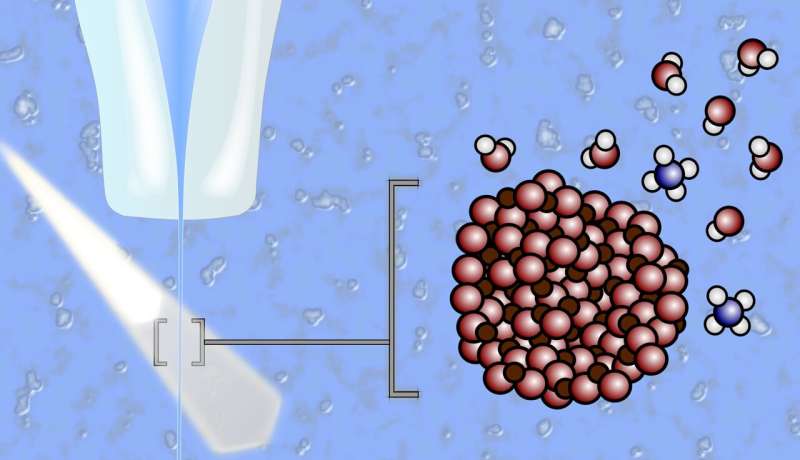
The liquid containing suspended metal-oxide nanoparticles explodes through X-ray light in a very thin jet. In this way, chemical reactions at the interfaces between solid metal-oxide particles and liquid electrolyte can be studied. Source: Robert Seidel / HZB
Green hydrogen can be produced directly in a photoelectrochemical cell, which splits water with solar energy. However, this requires the development of super-efficient photoelectrodes that must combine many talents at the same time: They must be very good at converting sunlight into electricity, remain stable in acidic or basic water, act as catalysts to promote the splitting of water into hydrogen and oxygen, and be cheap, abundant and non-toxic. The large material class of metal oxides is in question.
However, it is difficult to know exactly what happens at the interfaces between the solid metal oxide electrodes and the aqueous electrolyte. This is because standard X-ray analysis does not work to investigate the processes of samples in a liquid environment. One of the few suitable methods is experiments with a liquid jet: a fine liquid jet in which metal oxide nanoparticles are suspended.
This jet shoots through the X-ray light of BESSY II, and the interference of the evaporated molecules with the measurement data is negligible (more on the introduction of the special issue).
Dr. Robert Seidel is an expert in this liquid jet method, which is the subject of a special issue of Accounts of Chemical Research. He was invited to be a guest-editor of the issue and also to report on the new BESSY II experiments that he conducted with Dr. Hebatallah Ali and Dr. Bernd Winter from the Fritz Haber Institute.
They investigated two important model systems for photoelectrodes: Nanoparticles of iron oxide (hematite, α-Fe2or3and anatase (titanium oxide or TiO2) in aqueous electrolytes with different pH values. Hematite and anatase in suspension are photocatalytic model systems. It is ideal for studying the solid/electrolyte interface at the molecular level and for exploring chemical reactions at the electrode-electrolyte interface.
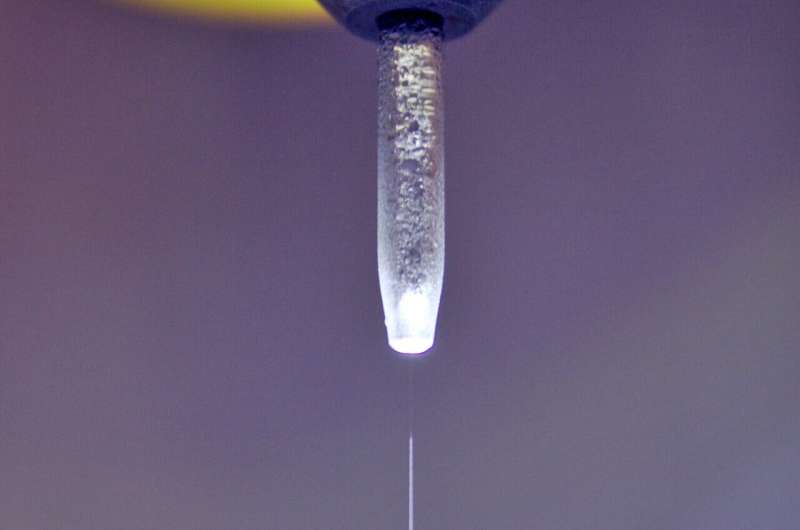
A microjet is a fast-flowing stream of liquid so narrow that it produces only a highly soluble vapor cloud. Photons and particles can reach and leave the surface of the jet without colliding with vapor molecules. Source: Robert Seidel / HZB
“We use resonant photoelectron spectroscopy (PES) to determine the characteristic fingerprints of various reactions. The key question: How do water molecules react on or on the surface of nanoparticles?
In fact, how acidic or how basic an electrolyte is makes a big difference, Seidel said. “At low pH, the water molecules on the hematite surface tend to dissociate. This is not the case with anatase, where the water molecules are adsorbed on the TiO surface.2 nanoparticles,” said Seidel. A basic pH value is required for water molecules to break down in anatase nanoparticles. “Such insights into the surface interactions of water molecules are possible only with this liquid-jet method,” said Seidel.
The spectra also revealed ultra-fast electron transitions between the metal oxide and the (split) surface water molecules. The results provide insights into the first steps of water dissociation and help elucidate the mechanisms of light-induced water splitting on metal oxide surfaces.
The special issue ‘Applications of Liquid Microjets in Chemistry’:
“Pour a glass of water, and bring it up to your nose, one centimeter away. What do you not see? About 3 million molecules intervene in a line between the tip of your nose and the surface of the water. Imagine an X-ray photon or charged or neutral particle trying to reach or escape the surface but first collides with some of these molecules interloping and interacting with the water about deep ways.”
What was so clearly described in the introduction to this special issue has long been a major problem. It was not until 1988 that the liquid jet method, introduced by Manfred Faubel, Stephan Schlemmer and Jan Peter Toennies, made it possible to study water surfaces without these disturbances. A microjet is a fast-flowing stream of liquid so narrow that it produces only a highly soluble vapor cloud. Photons and particles can reach and leave the surface of the jet without colliding with vapor molecules.
More information:
Hebatallah Ali et al, The Metal-Oxide Nanoparticle–Aqueous Solution Interface Studied by Liquid-Microjet Photoemission, Accounts of Chemical Research (2023). DOI: 10.1021/acs.accounts.2c00789
Provided by the Helmholtz Association of German Research Centers
Citation: BESSY II: Surface analysis of catalyst particles in aqueous solutions (2023, July 21) retrieved 21 July 2023 from https://phys.org/news/2023-07-bessy-ii-surface-analysis-catalyst.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
