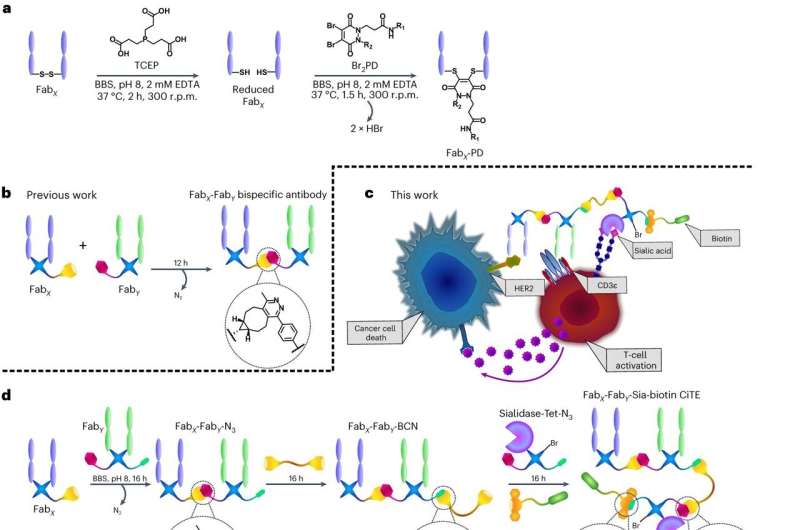
CiTE generation and proposed underlying biological mechanisms. a, The pyridazinedione method for the generation of functionalized Fabs. The Fab is first reduced by TCEP to liberate the cysteines of an interchain disulfide bond. The reduced Fab is then reacted with Br2The PD selected, through an addition-elimination mechanism in which thiols are successively transferred to each Br atom to create a strong covalent linkage between heavy and light protein chains. BBS, Borate buffered saline. bThe previously developed method for the generation of bsAbs with pyridazinediones by SPIEDAC click chemistry. c, Proposed mechanism of action of a CiTE-containing sialidase. CiTE binds to a target cancer cell through HER2-engagement and to a T cell through the CD3 co-receptor, crosslinking the two cells. The sialidase enzyme removes sialic acid from target and effector (T) cells to promote immune activation, leading to stronger T cell-mediated cytotoxicity. CiTE is functionalized with a biotin molecule to aid in imaging and/or purification. dThe method developed in this manuscript is for the generation of functionalized three-protein CiTE constructs. eThe pyridazinediones and other small molecules used in this work (Br2PD-Tet-N3 1BCN–PEG–BCN linker 2Bro2PD-BCN 3Bro2PD–Tue 4 and DBCO-biotin 5) for biotinylation of the constructs. Credit: Nature Chemistry (2023). DOI: 10.1038/s41557-023-01280-4
A powerful anti-cancer therapy has been developed using Nobel prize-winning “click chemistry,” where molecules click like LEGO bricks, in a new study by researchers at UCL and Stanford University.
The study, published in Nature Chemistryopens up new possibilities for how to develop cancer immunotherapies in the future.
The research team created an anti-cancer therapy with three components: one targeting the cancer cell, another recruiting white blood cells called T cells to attack the cancer cell, and the third knocking out part of the cancer cell’s defenses.
Previously, this type of three-component therapy was only done using a complex process called protein engineering, in which the DNA sequences for several proteins were combined and inserted into a single cell.
One of the three-part therapies the researchers developed, which uses an enzyme called sialidase to remove the sugars the cancer cell uses to hide itself, was particularly effective at killing breast cancer cells in a dish. The researchers say this shows that the enzyme—which has only recently begun to be explored in cancer research—has the potential to be the basis of next-generation anti-cancer agents.
The first author Dr. Peter Szijj (UCL Chemistry) said, “Click chemistry is a faster and more adaptable way to build these multifunctional anti-cancer agents than protein engineering. It is relatively easy to attach the click ‘handle’ to proteins so you can try many combinations quickly to test what works best. With protein engineering, you need a separate mechanism for each component.”
Senior corresponding author Professor Vijay Chudasama (UCL Chemistry) said, “As proteins are large and complex molecules, you need a combination of precise protein modification and reliable click chemistry to connect them in a controlled way. We have achieved this and show our strategy to be an interesting alternative to using classical protein engineering methods.”
“We hope that by using chemistry to create novel and highly sophisticated multi-protein anti-cancer agents we can encourage chemists to cross typical disciplinary boundaries to engage in novel applications in areas such as medical imaging, diagnostics and disease therapies.”
Click chemistry relies on two reaction partners (click handles) that can be attached to each other quickly and selectively, without producing any toxic products. These click handles can be added to proteins, in this case using functionalized pyridazinediones (PDs), allowing the proteins to click smoothly like LEGO.
Click chemistry pioneers have been awarded the 2022 Nobel Prize in Chemistry. Professor Carolyn R. Bertozzi, of Stanford University, who co-authored this latest paper, was one of three prize winners for her work on biorthogonal chemistry—click chemistry in living cells.
For the new paper, the UCL researchers first clicked two fragments of the antibody together—one fragment that binds to a cancer cell, another fragment that binds to a T cell so it can destroy the cancer cell. Similar T-cell engagers, created through protein engineering, have already been approved for use in humans and are being used to treat cancers such as multiple myeloma, a rare blood cancer, in the United States and Europe.
The team then added a third component, a checkpoint inhibitor, which removes an aspect of the cancer cell’s defenses. This substance is a PD-1-blocking antibody fragment, which is already used to treat certain advanced forms of skin or lung cancer and re-stimulates immune cells to target cancer cells; or the more experimental sialidase enzyme, which removes specific sugars (sialic acid) from the surface of the cancer cell as well as the T cell.
These sugars, present in all our cells, are produced in large numbers by cancer cells and help them hide from our immune system by turning off nearby immune cells.
The research team found that the addition of either of these components improved the effectiveness of the therapy in killing cancer, and that the addition of sialidase was even more powerful.
The researchers also added a fourth molecule, biotin, which allowed them to visualize how well the substances bound to their targets. They said it could be replaced by another small molecule with a different function—for example, to reduce side-effects by blocking the protein until it reaches its intended target: the cancer.
In the paper, the researchers say that using chemistry in this way to create cancer therapies shows “a lot of untapped potential that is still waiting to be discovered.”
This sialidase enzyme-containing therapeutic now needs to be tested on animals before any trials involving humans begin.
More information:
Peter A. Szijj et al, Chemical generation of checkpoint inhibitory T cell engagers for cancer treatment, Nature Chemistry (2023). DOI: 10.1038/s41557-023-01280-4
Provided by University College London
Citation: Potent anti-cancer therapy made using ‘click chemistry’ (2023, July 24) retrieved 24 July 2023 from https://phys.org/news/2023-07-potent-anti-cancer-therapy-click-chemistry.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
