
Researchers at the Institute for Basic Science (IBS) have confirmed the structure and properties of a transition metal-nitrenoid intermediate produced during catalytic amination reactions. Credit: Institute for Basic Science
Researchers at the Institute for Basic Science (IBS) in South Korea have made a groundbreaking discovery by identifying the structure and reactivity of rhodium-acylnitrenoid intermediates in catalytic hydrocarbon amination reactions. By studying the rhodium-dioxazolone coordination complex using photoinduced single crystal X-ray diffraction analysis, they obtained the transient moment of Rh-acylnitrenoid intermediate formation.
The findings are published in the journal Science.
This achievement is expected to pave the way for the production of highly reactive and selective catalysts for converting hydrocarbons into products with added value, with potential wide application in various industries.
Led by Director Chang Sukbok, scientists from the Center for Catalytic Hydrocarbon Functionalizations within the Institute for Basic Science (IBS) have made progress in understanding the structure and reactivity of an important intermediate in catalytic reactions. This intermediate, known as a transition metal-nitrenoid, plays an important role in converting hydrocarbons into amides, which are important in medicine and material science.
In chemical reactions, intermediates are substances that are formed and used during the transformation of reactants into products. Therefore, understanding these intermediates is important for improving reaction pathways and developing efficient catalysts. For example, nitrogen-containing compounds form the backbone of approximately 90% of pharmaceuticals and are essential in materials science.
Therefore, the identification of intermediates involved in amination reactions, where nitrogen-based functional groups are introduced to hydrocarbon raw materials, is very important.
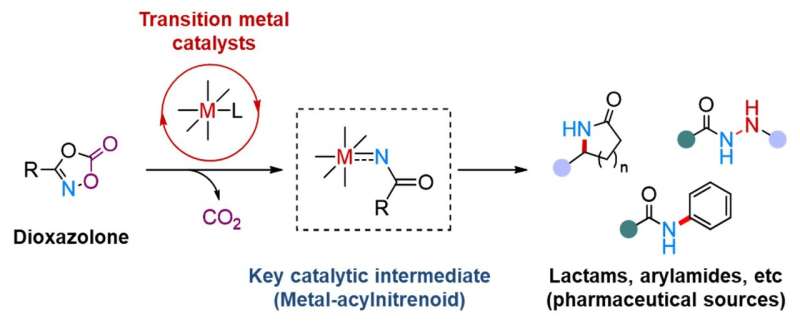
Metal-acylnitrenoid species are proposed as the key catalytic intermediates, leading to valuable nitrogen-containing molecules including lactams, and acrylamide, recognized as important scaffolds in pharmaceuticals and bioactive natural products. Credit: Institute for Basic Science
Researchers have recognized the importance of understanding the structure and properties of reaction intermediates in amination reactions. In particular, reactions using transition metal catalysts and dioxazolone reagents have been found to be very useful for medicinal chemistry and materials science, with more than 120 research groups worldwide contributing to the development of this field.
The key to understanding these reactions at a fundamental level lies in the ability to study the reaction intermediate that is formed when a transition-metal catalyst binds to a dioxazolone reagent—known as a metal-acylnitrenoid. These intermediate species are notoriously difficult to study because of their highly reactive nature, which only allows them to exist briefly.
In addition, traditional catalytic reactions usually take place in a solution, where intermediate substances react quickly with other molecules, which makes them more difficult to study.
To solve this challenge, the IBS team developed an experimental method using X-ray photocrystallography. In addition, they are also focused on tracking chemical reactions in solid-state rather than liquid solutions. For this purpose they developed a new chromophoric rhodium complex with a bidentate dioxazolone ligand, where photoinduced metal-to-ligand charge transfer initiates the catalytic CH amidation of hydrocarbon sources such as benzene.
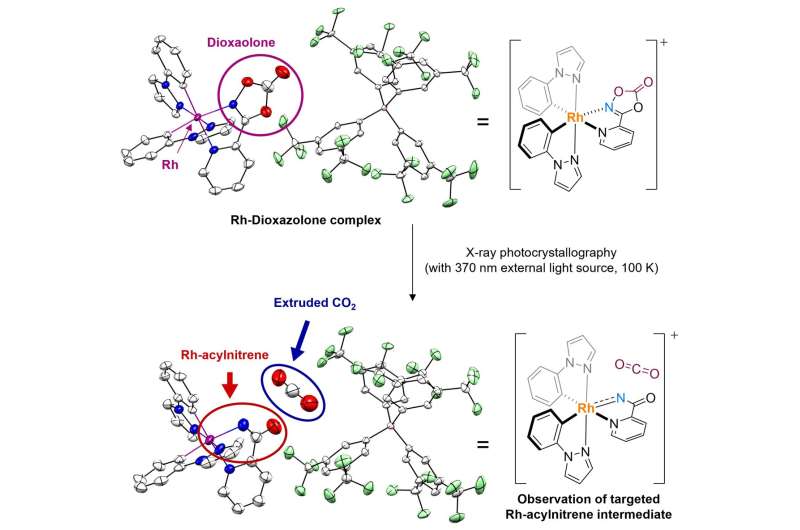
By using a crystal of the rhodium-bound dioxazolone coordination complex, the researchers observed the sought-after rhodium-acylnitrenoid species through photocrystallographic analysis. When dioxazolone reacts with transition metal catalysts to form metal-acylnitrenoids, one molecule of CO2 is extruded. Here, in the observed crystal structure, the CO2 molecule resides comfortably between the generated Rh-nitrenoid and the counter anion. Credit: Institute for Basic Science
Using this newly designed system, the researchers synthesized an isolable rhodium-dioxazolone coordination complex. Then, through photoinduced single crystal X-ray diffraction analysis using synchrotron radiation (Pohang Accelerator Laboratory), they were able to reveal the structure and properties of the rhodium-acylnitrenoid intermediate for the first time.
Furthermore, this study is designed to also achieve crystallographic monitoring of the transfer of rhodium-acylnitrene towards an external nucleophile all in the solid phase, providing complete mechanistic snapshots of the nitrenoid transfer process.
This research marks a significant step forward compared to previous research in the field of catalysis involving metal-nitrenoid intermediates. By observing metal-nitrenoid intermediates in catalytic reactions and the study provides significant insight into their reactivity. These findings are expected to contribute to the development of more reactive and selective catalysts for hydrocarbon amination reactions in the future.
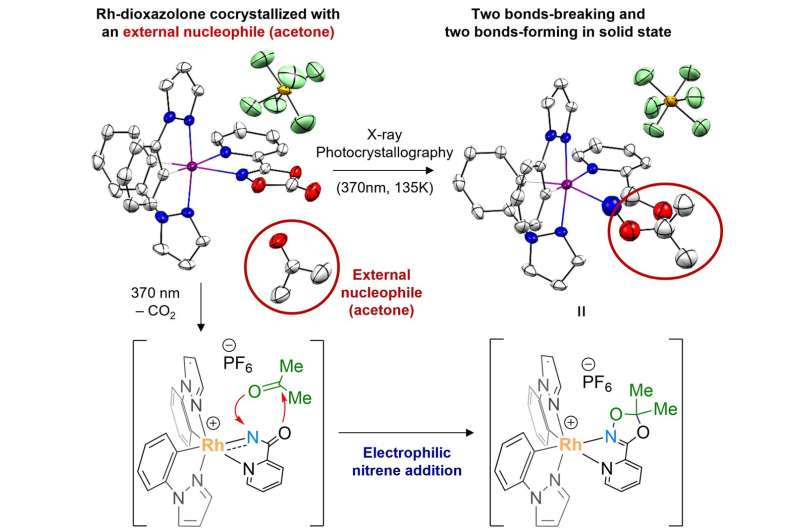
The researchers further prepared a cocrystal of rhodium-dioxazolone and a molecule of acetone, which allowed them to conduct additional photocrystallographic analysis to monitor the transfer of nitrenoid to a molecule of acetone as an external nucleophile. These results confirm the electrophilic reactivity nature of the rhodium-acylnitrenoid intermediate. Credit: Institute for Basic Science
Director Chang emphasized the importance of this discovery by saying, “We have experimentally obtained the transition metal-nitrenoid intermediate, whose existence was only hypothesized and difficult to prove.” He further noted that this research will provide important clues for the design of highly reactive and selective catalysts that will be useful in various industries, potentially contributing to the development of a “universal catalyst.”
More information:
Hoimin Jung et al, Mechanistic snapshots of rhodium-catalyzed acylnitrene transfer reactions, Science (2023). DOI: 10.1126/science.adh8753. www.science.org/doi/10.1126/science.adh8753
Provided by the Institute for Basic Science
Citation: Observation of a long postulated intermediate in catalytic amination reactions (2023, July 20) retrieved 20 July 2023 from https://phys.org/news/2023-07-long-postulated-intermediate-catalytic-amination-reactions.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
