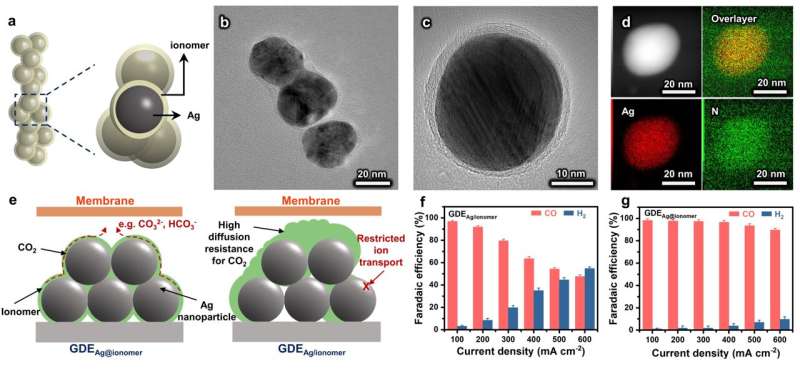
The ionomer is uniformly distributed around each catalyst particle. GDE using an ionomer-confined electrocatalyst optimizes the distribution of the ionomer within the catalyst layer, which not only establishes channels for the promoted transport of ions but also facilitates the mass transfer of CO.2. Source: Science China Press
Because the amount of carbon dioxide (CO2) in the atmosphere is increasing due to human activities, the rapid greenhouse effect and global warming are becoming common concerns. To reduce such environmental problems, various technologies have been developed to utilize CO2.
Electrocatalytic CO2 reducing reaction (CO2RR) is a very attractive CO conversion technology2 into value-added products using renewable energy (for example, solar and wind energy). To meet the requirements for industrial CO applications2RR, gas diffusion electrodes (GDEs)-based membrane electrode assemblies (MEAs) are widely used.
In GDE, the catalyst layer (CL) is the main site for the catalytic reaction, which usually consists of the catalyst, the catalyst carrier, and the ionomer. The preparation of CLs by ink-based methods usually suffers from inhomogeneous ionomer distribution, which affects the transport of ions and CO.2.
Therefore, rational optimization of the ionomer spatial distribution is useful to improve the mass transfer and improve the catalytic performance. In addition to methods such as adjusting the solvent polarity and improving the interaction between the catalyst and ionomer, there is still a need to develop a more proactive and controllable method to better optimize the ionomer distribution to achieve good performance for the practical use of CO.2RR systems.
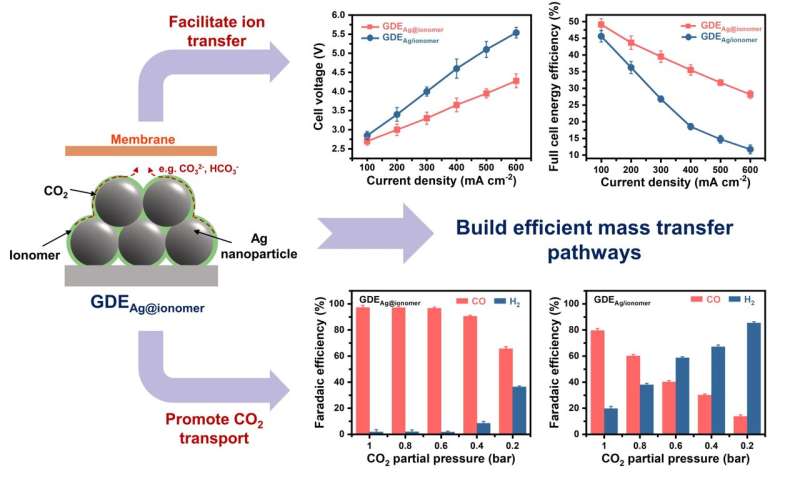
A pre-confinement method was developed to produce a gas diffusion electrode for electrochemical CO2 reduced reaction with uniformly distributed ionomer. The obtained electrode shows a low cell voltage of about 3.3 V at 300 mA cm−2 and a high CO Faradaic efficiency of more than 90% at 600 mA cm-2 due to the enhanced mass transfer of ions and reactants. Source: Science China Press
In response to this challenge, the Energy and Catalysis Adventure Team led by Professor Jinlong Gong from Tianjin University has developed an ionomer pre-confinement method. Specifically, the ionomer was introduced during the synthetic process of the electrocatalyst, which led to the formation of the ionomer-confined electrocatalyst for the construction of GDEs. This method favors the construction of GDEs with a uniform ionomer distribution, and then remits the mass transfer problems caused by its aggregation.
By optimizing the ionomer distribution in GDEs, ion transport within the CL is promoted. The ions generated at the reaction sites can be rapidly transferred to the anode. In addition, the spatially uniform ionomer avoids high local mass transfer resistance due to ionomer accumulation, which enhances CO.2 transport and improve catalytic performance.
Therefore, the optimized GDEs have a relatively low cell voltage (about 3.3 V at 300 mA cm-2) and a high CO Faradaic efficiency of more than 90% even at a high current density of 600 mA cm-2. This electrode also achieves stable catalysis with a current density of 300 mA cm-2 more than 220 hours. It is envisaged that the information gathered in this study will encourage the optimal design of GDEs, thus providing a reference for realizing the practical use of CO.2RR.
More information:
Xiaowei Du et al, Confinement of ionomer for electrocatalytic CO2 reduction reaction by efficient mass transfer pathways, National Science Review (2023). DOI: 10.1093/nsr/nwad149
Provided by Science China Press
Citation: Building efficient mass transfer pathways for the electrocatalytic carbon dioxide reduction reaction (2023, July 24) retrieved 24 July 2023 from https://phys.org/news/2023-07-efficient-mass-pathways-electrocatalytic-carbon.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
