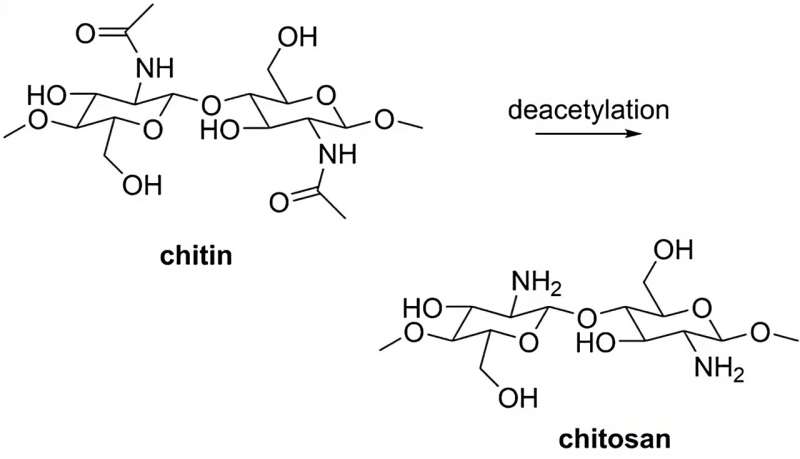
Chitosan is easily formed by deacetylation of chitin, which is the main component of insect skeletons and shrimp and crab shells. Credit: HIMS / HCSC.
Flexible spheres of the biomolecule chitosan, made from shrimp waste, can be used for catalysts that generate hydrogen gas from borohydride salts. In a paper on Green Chemistry, a research team at the University of Amsterdam (UvA) showed how the spheres can “breathe” hydrogen bubbles without bursting. This is an important step towards practical and safe hydrogen storage and release units.
Since 2020, the Heterogeneous Catalysis & Sustainable Chemistry group at the UvA’s Van ‘t Hoff Institute for Molecular Sciences has been working on the use of alkali metal borohydride salts as future hydrogen carriers. These solid salts can be safely stored in air under ambient conditions and release hydrogen gas only when reacting with water. However, preventing the release of hydrogen, and thereby preventing runaway reactions, is difficult.
One solution is to stabilize the solution with a base, and control the release of hydrogen by using a catalyst. The UvA team, led by Prof. Gadi Rothenberg, developed these catalysts in collaboration with the Austrian Competence Center for Tribology (AC2T) and the company Electriq Global.
Hydrogen destroys the catalyst particles
Finding potential catalysts is easy, but getting them to work long enough to be commercially viable is not. The combination of high reaction pH and continuous release of hydrogen bubbles destroys traditional catalysts within days. For example, the team succeeded in designing highly active and selective catalyst particles containing cobalt. High activity, however, results in high amounts of hydrogen that rapidly destroy particles.
The breakthrough came during the so-called Friday Afternoon experiment when MSc student Jeffrey Jonk and Ph.D. Student Fran Pope decided to try and encapsulate cobalt particles in chitosan spheres. Chitosan is a natural polymer made from chitin, the main component of insect exoskeletons and crustacean shells. It is a biodegradable, biocompatible material that is widely available on a multi-ton scale, mostly from shrimp and crab shell waste.
The repeating amine groups in the chitosan backbone make it highly soluble in aqueous acidic solutions but sparingly soluble in bases. Therefore, chitosan spheres are easily made by dropping liquid chitosan into a basic solution. An important property of chitosan spheres is their flexibility which enables them to expand during hydrogen production. Thus they can “breathe” hydrogen bubbles without bursting. And since they are made at high pH, the basicity of the borohydride solution is not a problem.
Real life potential for chitosan-based catalysts
The team tested the new catalysts in batch and continuous modes, monitoring the reactions by measuring the flow of hydrogen generated. A few mm-sized spheres filled with 7% cobalt were enough to generate 40 mL of hydrogen per minute in a continuous reactor for two days, demonstrating the real-life potential of this new catalyst.
According to Rothenberg the work emphasizes the importance of catalyst stability as a research focus. “Many papers are focused on activity and selection, because journals are focused on publishing unique results,” he said. “But if you look at the chemical industry, none of these ‘strange’ catalysts are used in practice. The reason is that running a successful reaction for a few hours, or even a few days, is meaningless for large-scale processes.
More information:
Frances Pope et al, From shrimp balls to hydrogen bubbles: borohydride hydrolysis catalysed by flexible cobalt chitosan spheres, Green Chemistry (2023). DOI: 10.1039/D3GC00821E
Given by the University of Amsterdam
Citation: Chemists recycle shrimp waste as catalyst for hydrogen generation (2023, July 20) retrieved 20 July 2023 from https://phys.org/news/2023-07-chemists-recycle-shrimp-catalyst-hydrogen.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
