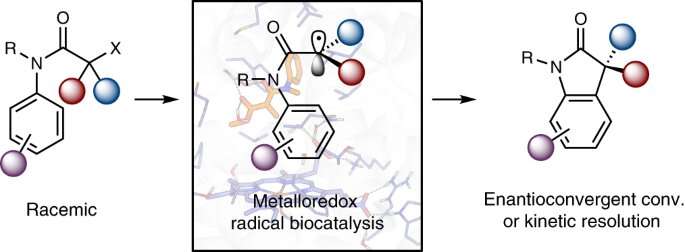
Graphic abstract. Credit: Catalysis in Nature (2023). DOI: 10.1038/s41929-023-00986-5
One of the central challenges for synthetic chemists is to control free radicals. Highly reactive molecules with an unpaired electron, free radicals, may be familiar to you; this is the type of molecule that we take antioxidant supplements for, in an effort to tame oxidative stress.
In the world of synthetic chemistry, however, free radicals hold great promise.
“Free radical chemistry is very useful for the synthesis of bioactive small molecules and everyday polymers,” said UC Santa Barbara chemistry professor Yang Yang, an author of a paper on the matter that appears in Catalysis in Nature. “However, imposing stereocontrol on free-radical mediated reactions has eluded the asymmetric catalysis community for decades. We are working to develop biocatalytic strategies to further push the boundaries of free radical chemistry.”
To fully unlock the synthetic potential of free radicals, Yang and colleagues focused on stereochemistry—also known as 3D chemistry, which focuses on the three-dimensional orientation of atoms and molecules.
The stereochemistry of organic molecules has a significant effect on their properties. For example, (S)-carvone or “left-handed” carvone is the main component responsible for the distinctive smell of mint. In contrast, (R)-carvone or “right-handed” carvone is found in caraway seeds and has a distinct odor. Thus, precise control of stereochemistry is a primary goal of synthetic chemistry. To achieve this, chemists turn to catalysts, substances that enable chemical reactions without being consumed or changed in the process, making them reusable.
Achieving this type of stereocontrol is no small feat. “In general, driving free radicals toward the desired stereochemistry is very difficult,” Yang said. Free radicals, once formed, do not interact well with the catalyst. In addition, these radicals are free in another sense—they are easily displaced from potential reactive sites.
But Yang and collaborators have some tricks up their sleeves: metalloenzymes – natural proteins with a reactive metal center, can create and prevent free radicals for selective changes.
“In particular in this paper we will solve a problem in this field, which is how to control the stereoselective addition of a radical species to an aromatic compound,” said Yang. “The radical in this case is derived from the racemic starting material.”
This is where three-dimensional chemistry comes in. “Racemic” means that the material is composed of equal proportions of “left-handed” and “right-handed” (also known as “chiral”) molecules—asymmetric molecules composed of the same atoms, so they are chemically the same, but are mirror images of each other. Like your hands, you can match them reflection-wise, but you can’t place them facing the same way. Which, under normal circumstances, would be a problem with enzymes.
“Enzymes are widely recognized as highly specific catalysts,” Yang said. “If enzymes are very specific, which happens often in nature, then the enzyme can only recognize and convert one enantiomeric form of a chiral compound,” Yang said. “And usually the enzyme doesn’t accept its mirror image.
“But in our work,” Yang continued, “we were able to create an enzyme that can accept both left-handed and right-handed forms of starting material, and then convert these starting materials to the same major enantiomeric product with excellent selectivity.”
In their paper, the researchers used an iron-dependent enzyme to produce reactive radical species. Through directed evolution, they engineered a set of selective iron enzymes to make either the left-handed or the right-handed product with excellent selectivity. Additionally, with a third, “kinetic resolution” enzyme, researchers can selectively transform the left-handed starting material, leaving the right-handed starting material untouched.
“So we have a toolbox of enzymes to allow different types of stereocontrol for the radical functionalization of aromatic compounds,” Yang said. “And yet these enzymes differ from each other by some mutation.” Yang hopes that this growing toolbox of biocatalysts will help others gain better control of their 3D chemistry, a classic problem that continues to confront organic chemists.
“Our metalloenzymes provide a potential general solution to control the selection of free radicals,” Yang said. “So these biocatalytic solutions we have developed are expected to facilitate the synthesis and study of chiral compounds in academia and industry.”
More information:
Wenzhen Fu et al, Enzyme-controlled stereoselective radical cyclization to arenes catalyzed by metalloredox biocatalysis, Catalysis in Nature (2023). DOI: 10.1038/s41929-023-00986-5
Provided by the University of California – Santa Barbara
Citation: A ‘toolbox of biocatalysts’ improves control of free radicals (2023, July 21) retrieved 21 July 2023 from https://phys.org/news/2023-07-toolbox-biocatalysts-free-radicals.html
This document is subject to copyright. Except for any fair dealing for the purpose of private study or research, no part may be reproduced without written permission. Content is provided for informational purposes only.
